| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2007-05-22 21:57:36 UTC |
|---|
| Update Date | 2022-03-07 02:49:31 UTC |
|---|
| HMDB ID | HMDB0006292 |
|---|
| Secondary Accession Numbers | - HMDB0002325
- HMDB02325
- HMDB06292
|
|---|
| Metabolite Identification |
|---|
| Common Name | Chenodeoxycholoyl-CoA |
|---|
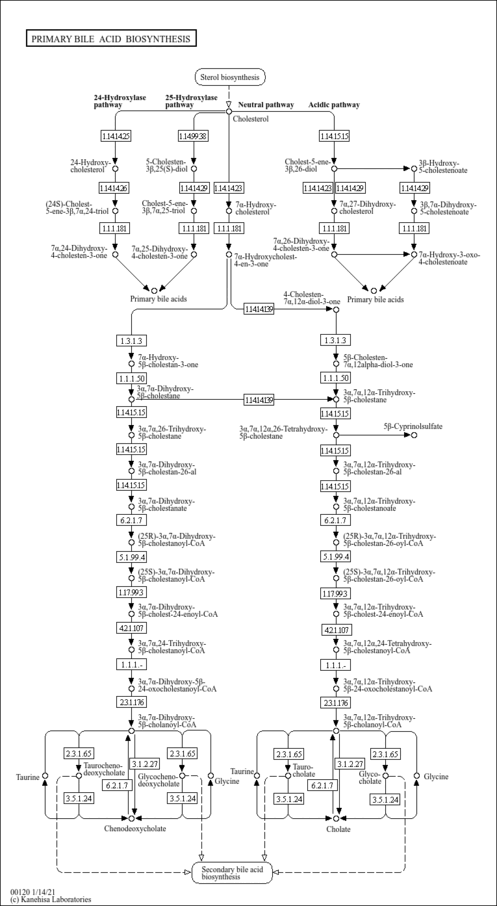
| Description | Chenodeoxycholoyl-CoA is bile acid Coenzyme A ester. In humans, bile acids conjugated with glycine and taurine are the major solutes in bile, and unconjugated bile acids are almost nondetectable in normal bile. Conjugated bile acids are less toxic and are more efficient promoters of intestinal absorption of dietary lipid than unconjugated bile acids. The synthesis of bile acid and amino acid conjugates in human liver is the result of two independent enzymatic reactions with a bile acid coenzyme A thioester intermediate formation of bile acid-CoA esters, considered the rate-limiting step in bile acid amidation and catalyzed by an ATP-dependent microsomal enzyme, bile acid-CoA synthetase (EC 6.2.1.7). In the second reaction, the thioester bond is cleaved, and an amide bond is formed between the bile acid and the amino acids glycine or taurine. The bile acid-CoA:amino acid N-acyltransferase (EC 2.3.1.65) catalyzes this reaction in the cytosol prior to secretion into bile. In human liver the formation of bile acid-CoA thioesters is localized both to the microsomal fraction catalysed by an ATP-dependent synthetase and to the peroxisomal fraction catalysed by the thiolase in the last step of the beta-oxidative cleavage of the 5beta-cholestanoyl side chain. The highest specific amidation activity of both chenodeoxycholoyl-CoA is always found in the most peroxisome-rich subcellular fractions. (PMID: 2722825 , 10817395 , 11673457 , 10884298 ). |
|---|
| Structure | [H][C@@]1(CC[C@@]2([H])[C@]3([H])[C@H](O)C[C@]4([H])C[C@H](O)CC[C@]4(C)[C@@]3([H])CC[C@]12C)[C@H](C)CCC(=O)SCCNC(=O)CCNC(=O)C(O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)N1C=NC2=C1N=CN=C2N InChI=1S/C45H74N7O19P3S/c1-24(27-7-8-28-34-29(11-14-45(27,28)5)44(4)13-10-26(53)18-25(44)19-30(34)54)6-9-33(56)75-17-16-47-32(55)12-15-48-41(59)38(58)43(2,3)21-68-74(65,66)71-73(63,64)67-20-31-37(70-72(60,61)62)36(57)42(69-31)52-23-51-35-39(46)49-22-50-40(35)52/h22-31,34,36-38,42,53-54,57-58H,6-21H2,1-5H3,(H,47,55)(H,48,59)(H,63,64)(H,65,66)(H2,46,49,50)(H2,60,61,62)/t24-,25+,26-,27-,28+,29+,30-,31-,34+,36-,37-,38?,42-,44+,45-/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| 5'-{3-[(3R)-4-{[3-({2-[(3alpha,7alpha-dihydroxy-5beta-cholan-24-oyl)sulfanyl]ethyl}amino)-3-oxopropyl]amino}-3-hydroxy-2,2-dimethyl-4-oxobutyl] dihydrogen diphosphate}3'-phosphoadenosine | HMDB | | Chenodeoxycholoyl coenzyme A | HMDB |
|
|---|
| Chemical Formula | C45H74N7O19P3S |
|---|
| Average Molecular Weight | 1142.091 |
|---|
| Monoisotopic Molecular Weight | 1141.397303447 |
|---|
| IUPAC Name | {[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-2-({[({[3-({2-[(2-{[(4R)-4-[(1S,2S,5R,7S,9R,10R,11S,14R,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]pentanoyl]sulfanyl}ethyl)carbamoyl]ethyl}carbamoyl)-3-hydroxy-2,2-dimethylpropoxy](hydroxy)phosphoryl}oxy)(hydroxy)phosphoryl]oxy}methyl)-4-hydroxyoxolan-3-yl]oxy}phosphonic acid |
|---|
| Traditional Name | [(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-2-[({[3-({2-[(2-{[(4R)-4-[(1S,2S,5R,7S,9R,10R,11S,14R,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]pentanoyl]sulfanyl}ethyl)carbamoyl]ethyl}carbamoyl)-3-hydroxy-2,2-dimethylpropoxy(hydroxy)phosphoryl]oxy(hydroxy)phosphoryl}oxy)methyl]-4-hydroxyoxolan-3-yl]oxyphosphonic acid |
|---|
| CAS Registry Number | 60731-52-4 |
|---|
| SMILES | [H][C@@]1(CC[C@@]2([H])[C@]3([H])[C@H](O)C[C@]4([H])C[C@H](O)CC[C@]4(C)[C@@]3([H])CC[C@]12C)[C@H](C)CCC(=O)SCCNC(=O)CCNC(=O)C(O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)N1C=NC2=C1N=CN=C2N |
|---|
| InChI Identifier | InChI=1S/C45H74N7O19P3S/c1-24(27-7-8-28-34-29(11-14-45(27,28)5)44(4)13-10-26(53)18-25(44)19-30(34)54)6-9-33(56)75-17-16-47-32(55)12-15-48-41(59)38(58)43(2,3)21-68-74(65,66)71-73(63,64)67-20-31-37(70-72(60,61)62)36(57)42(69-31)52-23-51-35-39(46)49-22-50-40(35)52/h22-31,34,36-38,42,53-54,57-58H,6-21H2,1-5H3,(H,47,55)(H,48,59)(H,63,64)(H,65,66)(H2,46,49,50)(H2,60,61,62)/t24-,25+,26-,27-,28+,29+,30-,31-,34+,36-,37-,38?,42-,44+,45-/m1/s1 |
|---|
| InChI Key | IIWDDMINEZBCTG-POZCYTSJSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as 2,3,4-saturated fatty acyl coas. These are acyl-CoAs carrying a 2,3,4-saturated fatty acyl chain. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Fatty acyl thioesters |
|---|
| Direct Parent | 2,3,4-saturated fatty acyl CoAs |
|---|
| Alternative Parents | |
|---|
| Substituents | - Coenzyme a or derivatives
- Purine ribonucleoside 3',5'-bisphosphate
- Purine ribonucleoside bisphosphate
- Purine ribonucleoside diphosphate
- Steroidal glycoside
- Dihydroxy bile acid, alcohol, or derivatives
- Hydroxy bile acid, alcohol, or derivatives
- Cholane-skeleton
- Bile acid, alcohol, or derivatives
- 3-hydroxysteroid
- Hydroxysteroid
- 3-alpha-hydroxysteroid
- 7-hydroxysteroid
- Steroid
- Ribonucleoside 3'-phosphate
- Pentose phosphate
- Pentose-5-phosphate
- Beta amino acid or derivatives
- Glycosyl compound
- N-glycosyl compound
- Organic pyrophosphate
- 6-aminopurine
- Pentose monosaccharide
- Monosaccharide phosphate
- Purine
- Imidazopyrimidine
- Monoalkyl phosphate
- Aminopyrimidine
- Pyrimidine
- Monosaccharide
- N-acyl-amine
- Phosphoric acid ester
- Fatty amide
- N-substituted imidazole
- Imidolactam
- Organic phosphoric acid derivative
- Alkyl phosphate
- Azole
- Tetrahydrofuran
- Cyclic alcohol
- Imidazole
- Heteroaromatic compound
- Amino acid or derivatives
- Carbothioic s-ester
- Carboxamide group
- Thiocarboxylic acid ester
- Secondary carboxylic acid amide
- Secondary alcohol
- Thiocarboxylic acid or derivatives
- Oxacycle
- Sulfenyl compound
- Azacycle
- Carboxylic acid derivative
- Organoheterocyclic compound
- Primary amine
- Alcohol
- Carbonyl group
- Organic oxygen compound
- Organosulfur compound
- Organooxygen compound
- Organopnictogen compound
- Amine
- Organic nitrogen compound
- Organonitrogen compound
- Hydrocarbon derivative
- Organic oxide
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | |
|---|
| Physical Properties |
|---|
| State | Gas |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | 1.41 | HANSCH,C ET AL. (1995) |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesNot Available |
|---|
| MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 10V, Positive-QTOF | splash10-000i-1900111000-49f063c429232551ee17 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 20V, Positive-QTOF | splash10-000i-0900215000-8d2226be8c0c4bc5d849 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 40V, Positive-QTOF | splash10-000i-1900001000-7a0a8e03b96562805e53 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 10V, Negative-QTOF | splash10-00al-2902140300-27178d920f5623fe9229 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 20V, Negative-QTOF | splash10-001i-3901120100-6b56cd2f9088ae3e0e8e | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 40V, Negative-QTOF | splash10-057i-5900100000-5f68bcc57183d55aa9fc | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 10V, Negative-QTOF | splash10-0006-0900000000-f2029e50b149beb9f9f2 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 20V, Negative-QTOF | splash10-006x-2901201100-35d0be690cfa669a6345 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 40V, Negative-QTOF | splash10-00p3-7904802600-c387954862814720775d | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 10V, Positive-QTOF | splash10-006x-0900000000-46620c2f6efcaebe68a6 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 20V, Positive-QTOF | splash10-0079-2900000011-05d1a4743e5cbc0dc9bb | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Chenodeoxycholoyl-CoA 40V, Positive-QTOF | splash10-000i-0000019000-4efd57469a29c0b4117b | 2021-09-24 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum |
|
|---|