| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2023-02-21 17:14:40 UTC |
|---|
| HMDB ID | HMDB0000263 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Phosphoenolpyruvic acid |
|---|
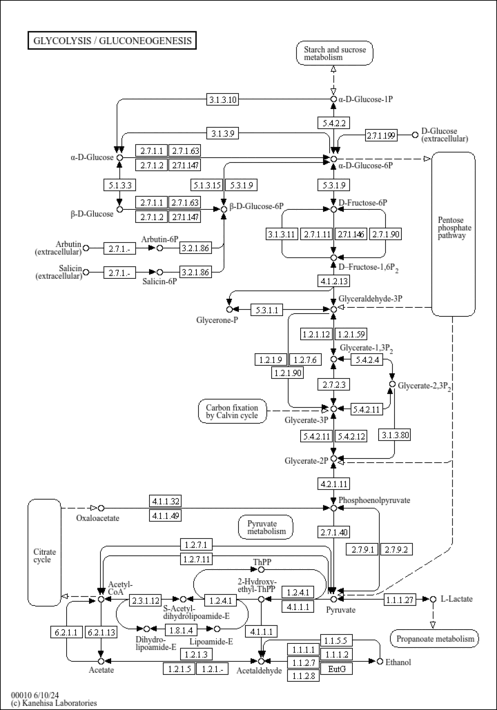
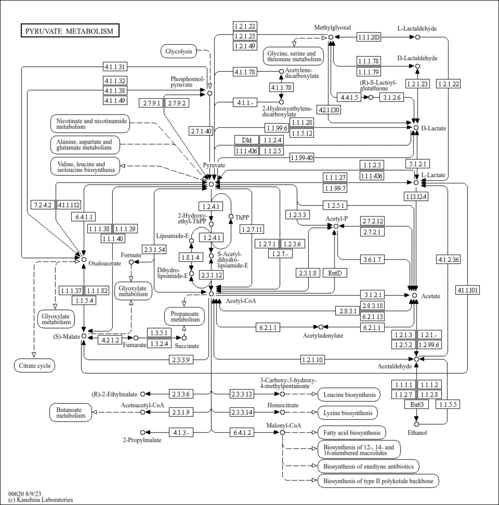
| Description | Phosphoenolpyruvate (PEP) is an important chemical compound in biochemistry. It has a high energy phosphate bond, and is involved in glycolysis and gluconeogenesis. In glycolysis, PEP is formed by the action of the enzyme enolase on 2-phosphoglycerate. Metabolism of PEP to pyruvate by pyruvate kinase (PK) generates 1 molecule of adenosine triphosphate (ATP) via substrate-level phosphorylation. ATP is one of the major currencies of chemical energy within cells. In gluconeogenesis, PEP is formed from the decarboxylation of oxaloacetate and hydrolysis of 1 guanosine triphosphate molecule. This reaction is catalyzed by the enzyme phosphoenolpyruvate carboxykinase (PEPCK). This reaction is a rate-limiting step in gluconeogenesis. (wikipedia). |
|---|
| Structure | InChI=1S/C3H5O6P/c1-2(3(4)5)9-10(6,7)8/h1H2,(H,4,5)(H2,6,7,8) |
|---|
| Synonyms | | Value | Source |
|---|
| 2-(Phosphonooxy)-2-propenoic acid | ChEBI | | 2-PHOSPHOENOLPYRUVIC ACID | ChEBI | | PEP | ChEBI | | PHOSPHOENOLPYRUVATE | ChEBI | | 2-(Phosphonooxy)-2-propenoate | Generator | | 2-PHOSPHOENOLPYRUVate | Generator | | 2-Hydroxy-acrylic acid dihydrogen phosphate | HMDB | | 2-Phosphonooxyprop-2-enoate | HMDB | | 2-Phosphonooxyprop-2-enoic acid | HMDB | | p-enol-Pyruvate | HMDB | | PEP (phosphate) | HMDB | | Phosphoenolpyruvic acid | HMDB |
|
|---|
| Chemical Formula | C3H5O6P |
|---|
| Average Molecular Weight | 168.042 |
|---|
| Monoisotopic Molecular Weight | 167.982374404 |
|---|
| IUPAC Name | 2-(phosphonooxy)prop-2-enoic acid |
|---|
| Traditional Name | phosphoenolpyruvic acid |
|---|
| CAS Registry Number | 138-08-9 |
|---|
| SMILES | OC(=O)C(=C)OP(O)(O)=O |
|---|
| InChI Identifier | InChI=1S/C3H5O6P/c1-2(3(4)5)9-10(6,7)8/h1H2,(H,4,5)(H2,6,7,8) |
|---|
| InChI Key | DTBNBXWJWCWCIK-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as phosphate esters. These are organic compounds containing phosphoric acid ester functional group, with the general structure R1P(=O)(R2)OR3. R1,R2 = O,N, or halogen atom; R3 = organyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Organic phosphoric acids and derivatives |
|---|
| Sub Class | Phosphate esters |
|---|
| Direct Parent | Phosphate esters |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phosphoric acid ester
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Experimental Collision Cross Sections |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| Phosphoenolpyruvic acid,1TMS,isomer #1 | C=C(OP(=O)(O)O)C(=O)O[Si](C)(C)C | 1466.6 | Semi standard non polar | 33892256 | | Phosphoenolpyruvic acid,1TMS,isomer #2 | C=C(OP(=O)(O)O[Si](C)(C)C)C(=O)O | 1478.4 | Semi standard non polar | 33892256 | | Phosphoenolpyruvic acid,2TMS,isomer #1 | C=C(OP(=O)(O)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1530.4 | Semi standard non polar | 33892256 | | Phosphoenolpyruvic acid,2TMS,isomer #1 | C=C(OP(=O)(O)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1546.5 | Standard non polar | 33892256 | | Phosphoenolpyruvic acid,2TMS,isomer #1 | C=C(OP(=O)(O)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1930.4 | Standard polar | 33892256 | | Phosphoenolpyruvic acid,2TMS,isomer #2 | C=C(OP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)C(=O)O | 1564.6 | Semi standard non polar | 33892256 | | Phosphoenolpyruvic acid,2TMS,isomer #2 | C=C(OP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)C(=O)O | 1542.3 | Standard non polar | 33892256 | | Phosphoenolpyruvic acid,2TMS,isomer #2 | C=C(OP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)C(=O)O | 1819.8 | Standard polar | 33892256 | | Phosphoenolpyruvic acid,3TMS,isomer #1 | C=C(OP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1615.7 | Semi standard non polar | 33892256 | | Phosphoenolpyruvic acid,3TMS,isomer #1 | C=C(OP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1606.9 | Standard non polar | 33892256 | | Phosphoenolpyruvic acid,3TMS,isomer #1 | C=C(OP(=O)(O[Si](C)(C)C)O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1718.0 | Standard polar | 33892256 | | Phosphoenolpyruvic acid,1TBDMS,isomer #1 | C=C(OP(=O)(O)O)C(=O)O[Si](C)(C)C(C)(C)C | 1705.6 | Semi standard non polar | 33892256 | | Phosphoenolpyruvic acid,1TBDMS,isomer #2 | C=C(OP(=O)(O)O[Si](C)(C)C(C)(C)C)C(=O)O | 1732.5 | Semi standard non polar | 33892256 | | Phosphoenolpyruvic acid,2TBDMS,isomer #1 | C=C(OP(=O)(O)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 1966.1 | Semi standard non polar | 33892256 | | Phosphoenolpyruvic acid,2TBDMS,isomer #1 | C=C(OP(=O)(O)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 1933.9 | Standard non polar | 33892256 | | Phosphoenolpyruvic acid,2TBDMS,isomer #1 | C=C(OP(=O)(O)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 2197.6 | Standard polar | 33892256 | | Phosphoenolpyruvic acid,2TBDMS,isomer #2 | C=C(OP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)C(=O)O | 1982.0 | Semi standard non polar | 33892256 | | Phosphoenolpyruvic acid,2TBDMS,isomer #2 | C=C(OP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)C(=O)O | 1950.9 | Standard non polar | 33892256 | | Phosphoenolpyruvic acid,2TBDMS,isomer #2 | C=C(OP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)C(=O)O | 2084.2 | Standard polar | 33892256 | | Phosphoenolpyruvic acid,3TBDMS,isomer #1 | C=C(OP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 2198.1 | Semi standard non polar | 33892256 | | Phosphoenolpyruvic acid,3TBDMS,isomer #1 | C=C(OP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 2144.2 | Standard non polar | 33892256 | | Phosphoenolpyruvic acid,3TBDMS,isomer #1 | C=C(OP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 2102.7 | Standard polar | 33892256 |
|
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental GC-MS | GC-MS Spectrum - Phosphoenolpyruvic acid GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (Non-derivatized) | splash10-0292-0962000000-44d3914b4e07e5c50e3f | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - Phosphoenolpyruvic acid GC-EI-TOF (Non-derivatized) | splash10-0292-0962000000-44d3914b4e07e5c50e3f | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Phosphoenolpyruvic acid GC-MS (Non-derivatized) - 70eV, Positive | splash10-0002-9200000000-ae9d81fc88a98f678268 | 2016-09-22 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Phosphoenolpyruvic acid GC-MS (1 TMS) - 70eV, Positive | splash10-00di-9410000000-072478606f0ab6809902 | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Phosphoenolpyruvic acid GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Phosphoenolpyruvic acid GC-MS (TMS_1_2) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Phosphoenolpyruvic acid GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Phosphoenolpyruvic acid GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid Quattro_QQQ 10V, Positive-QTOF (Annotated) | splash10-0udr-1900000000-9a4f5554af9a717d99c7 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid Quattro_QQQ 25V, Positive-QTOF (Annotated) | splash10-000x-9200000000-6cdc15f3daa25fc41655 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid Quattro_QQQ 40V, Positive-QTOF (Annotated) | splash10-001i-9100000000-39e22409d6d924a774f1 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-00xu-0912000000-ad1823470675975d5ff0 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-0udi-0900000000-1f5b761ce5fa374b0f8e | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-000i-9000000000-d279f0ca2accb130181f | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-0002-0920000000-f084dccf78e11c8760d7 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-00lr-0911000000-7fa833698210746c838f | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-000i-9000000000-62e28301f20b08971b78 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-0udi-0900000000-25f970898112f7b89898 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive-QTOF | splash10-0002-0930000000-58691b23d317c2418c33 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-00p0-0493110000-9d61bb13ab5261fdf9fd | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-004i-9100000000-db409b3cfa9ebabbcb58 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-0a4j-5090000000-1ee56bc4866301d4bf2e | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative-QTOF | splash10-0a4i-0090000000-bce08b01d44391bfecad | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative-QTOF | splash10-016r-7900000000-ffd3b8dcd65aaea26ac3 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative-QTOF | splash10-004i-9000000000-ceae3587b1e7d8b3da27 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative-QTOF | splash10-004i-9000000000-701a17330cd18255d625 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative-QTOF | splash10-004i-9000000000-a4178ce4951e2c3dba7b | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-QQ (API3000, Applied Biosystems) 50V, Negative-QTOF | splash10-004i-9000000000-9d0421620a7aaa9ef33f | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-QQ (API3000, Applied Biosystems) 10V, Positive-QTOF | splash10-0k9i-1900000000-fb12ade375a7ac97f150 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-QQ (API3000, Applied Biosystems) 20V, Positive-QTOF | splash10-0f76-8900000000-7f57e6a8e72e5992cd88 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-QQ (API3000, Applied Biosystems) 30V, Positive-QTOF | splash10-000j-9300000000-c02bca019150d572b3bc | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-QQ (API3000, Applied Biosystems) 40V, Positive-QTOF | splash10-05bf-9200000000-73fa5ca52ecc17bac380 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Phosphoenolpyruvic acid LC-ESI-QQ (API3000, Applied Biosystems) 50V, Positive-QTOF | splash10-0079-9300000000-231346190165f44f28dc | 2012-08-31 | HMDB team, MONA | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Experimental 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, experimental) | 2012-12-04 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Experimental 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | 2012-12-05 | Wishart Lab | View Spectrum |
IR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M-H]-) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+H]+) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+Na]+) | 2023-02-03 | FELIX lab | View Spectrum |
|
|---|
| General References | - Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. doi: 10.1038/nature07762. [PubMed:19212411 ]
- Nakayama Y, Kinoshita A, Tomita M: Dynamic simulation of red blood cell metabolism and its application to the analysis of a pathological condition. Theor Biol Med Model. 2005 May 9;2:18. [PubMed:15882454 ]
- Krogh P: Role of ochratoxin in disease causation. Food Chem Toxicol. 1992 Mar;30(3):213-24. [PubMed:1618445 ]
- Germaine GR, Tellefson LM: Promotion of Streptococcus mutans glucose transport by human whole saliva and parotid fluid. Infect Immun. 1985 Apr;48(1):7-13. [PubMed:3980096 ]
- Schatzberger P: Maternity services. BMJ. 1992 May 23;304(6838):1382-3. [PubMed:1611358 ]
- Orye E, Verhaaren H, Samuel K, van Mele B: A 46,XX,10Q+ chromosome constitution in a girl. Partial long arm duplication or insertional translocation? Humangenetik. 1975 May 26;28(1):1-8. [PubMed:1150258 ]
- Landau BR, Chandramouli V, Schumann WC, Ekberg K, Kumaran K, Kalhan SC, Wahren J: Estimates of Krebs cycle activity and contributions of gluconeogenesis to hepatic glucose production in fasting healthy subjects and IDDM patients. Diabetologia. 1995 Jul;38(7):831-8. [PubMed:7556986 ]
- Shirokane Y, Nakajima M, Mizusawa K: A new enzymatic assay of urinary guanidinoacetic acid. Clin Chim Acta. 1991 Oct 31;202(3):227-36. [PubMed:1667626 ]
- Tannen RL: Ammonia metabolism. Am J Physiol. 1978 Oct;235(4):F265-77. [PubMed:29492 ]
- Atkin BM, Buist NR, Utter MF, Leiter AB, Banker BQ: Pyruvate carboxylase deficiency and lactic acidosis in a retarded child without Leigh's disease. Pediatr Res. 1979 Feb;13(2):109-16. [PubMed:219411 ]
- Bojarska-Dahlig H, Gloabski T, Dzioegielewska I: [Salts of cyclic erythromycin A carbonate with cinnamic acid derivatives]. Acta Pol Pharm. 1975;32(3):311-7. [PubMed:1155186 ]
- Matsumoto T, van der Auwera P, Watanabe Y, Tanaka M, Ogata N, Naito S, Kumazawa J: Neutrophil function in hyperosmotic NaCl is preserved by phosphoenol pyruvate. Urol Res. 1991;19(4):223-7. [PubMed:1656579 ]
- Cahill GF Jr, Aoki TT: Renal gluconeogenesis and amino-acid metabolism in man. Med Clin North Am. 1975 May;59(3):751-61. [PubMed:1092934 ]
- Beyer C: Creatine measurement in serum and urine with an automated enzymatic method. Clin Chem. 1993 Aug;39(8):1613-9. [PubMed:8353946 ]
- Momeni N, Yoshimoto T, Ryberg B, Sandberg-Wollheim M, Grubb A: Factors influencing analysis of prolyl endopeptidase in human blood and cerebrospinal fluid: increase in assay sensitivity. Scand J Clin Lab Invest. 2003;63(6):387-95. [PubMed:14594319 ]
- Elshenawy S, Pinney SE, Stuart T, Doulias PT, Zura G, Parry S, Elovitz MA, Bennett MJ, Bansal A, Strauss JF 3rd, Ischiropoulos H, Simmons RA: The Metabolomic Signature of the Placenta in Spontaneous Preterm Birth. Int J Mol Sci. 2020 Feb 4;21(3). pii: ijms21031043. doi: 10.3390/ijms21031043. [PubMed:32033212 ]
|
|---|