| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2006-08-14 00:42:32 UTC |
|---|
| Update Date | 2022-03-07 02:49:21 UTC |
|---|
| HMDB ID | HMDB0004694 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | 11,14,15-THETA |
|---|
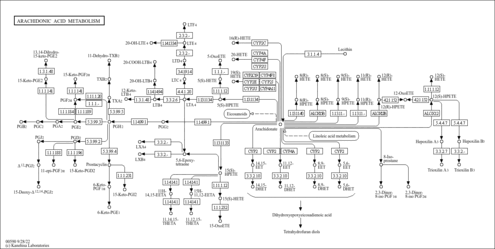
| Description | 11,14,15-trihydroxyeicosatrienoic acid (11,14,15-THETA) is a metabolite of the 15-lipoxygenase (15-LO) pathway of arachidonic acid (AA). Increased amounts of 11,14,15-THETA are synthesized in subacute hypoxia. Prolonged exposure to reduced PO2 activates 15-LO in small pulmonary arteries (PA); activation of 15-LO is associated with translocation of the enzyme from the cytosol to membrane. 11,14,15-THETA is an endothelium-derived relaxing factor. (PMID: 12690037 , 9812980 , 15388505 , 14622984 ). |
|---|
| Structure | CCCCCC(O)C(O)\C=C\C(O)C\C=C/C\C=C/CCCC(O)=O InChI=1S/C20H34O5/c1-2-3-9-13-18(22)19(23)16-15-17(21)12-10-7-5-4-6-8-11-14-20(24)25/h4,6-7,10,15-19,21-23H,2-3,5,8-9,11-14H2,1H3,(H,24,25)/b6-4-,10-7-,16-15+ |
|---|
| Synonyms | | Value | Source |
|---|
| (5Z,8Z,12E)-11,14,15-Trihydroxyeicosa-5,8,12-trienoic acid | ChEBI | | (5Z,8Z,12E)-11,14,15-Trihydroxyeicosatrienoic acid | ChEBI | | (5Z,8Z,12E)-11,14,15-Trihydroxyicosatrienoic acid | ChEBI | | 11,14,15-Trihydroxy-(5Z,8Z,12E)-eicosatrienoic acid | ChEBI | | 11,14,15-Trihydroxy-5Z,8Z,12E-eicosatrienoic acid | ChEBI | | 11,14,15-Trihydroxyicosatrienoic acid | ChEBI | | (5Z,8Z,12E)-11,14,15-Trihydroxyicosa-5,8,12-trienoic acid | Kegg | | (5Z,8Z,12E)-11,14,15-Trihydroxyeicosa-5,8,12-trienoate | Generator | | (5Z,8Z,12E)-11,14,15-Trihydroxyeicosatrienoate | Generator | | (5Z,8Z,12E)-11,14,15-Trihydroxyicosatrienoate | Generator | | 11,14,15-Trihydroxy-(5Z,8Z,12E)-eicosatrienoate | Generator | | 11,14,15-Trihydroxy-5Z,8Z,12E-eicosatrienoate | Generator | | 11,14,15-Trihydroxyicosatrienoate | Generator | | (5Z,8Z,12E)-11,14,15-Trihydroxyicosa-5,8,12-trienoate | Generator | | 11,14,15-THET | HMDB | | 11,14,15-Trihydroxyeicosa-5,8,12-trienoic acid | HMDB | | 11,14,15-Trihydroxyeicosatetraenoic acid | HMDB |
|
|---|
| Chemical Formula | C20H34O5 |
|---|
| Average Molecular Weight | 354.481 |
|---|
| Monoisotopic Molecular Weight | 354.240624198 |
|---|
| IUPAC Name | (5Z,8Z,12E)-11,14,15-trihydroxyicosa-5,8,12-trienoic acid |
|---|
| Traditional Name | (5Z,8Z,12E)-11,14,15-trihydroxyicosa-5,8,12-trienoic acid |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | CCCCCC(O)C(O)\C=C\C(O)C\C=C/C\C=C/CCCC(O)=O |
|---|
| InChI Identifier | InChI=1S/C20H34O5/c1-2-3-9-13-18(22)19(23)16-15-17(21)12-10-7-5-4-6-8-11-14-20(24)25/h4,6-7,10,15-19,21-23H,2-3,5,8-9,11-14H2,1H3,(H,24,25)/b6-4-,10-7-,16-15+ |
|---|
| InChI Key | YCFPVUKKIWMCJK-LTCHCNGXSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as hydroxyeicosatrienoic acids. These are eicosanoic acids with an attached hydroxyl group and three CC double bonds. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Eicosanoids |
|---|
| Direct Parent | Hydroxyeicosatrienoic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Hydroxyeicosatrienoic acid
- Long-chain fatty acid
- Hydroxy fatty acid
- Fatty acid
- Unsaturated fatty acid
- Secondary alcohol
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Polyol
- Organic oxide
- Organic oxygen compound
- Hydrocarbon derivative
- Carbonyl group
- Organooxygen compound
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| 11,14,15-THETA,1TMS,isomer #1 | CCCCCC(O[Si](C)(C)C)C(O)/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O | 2990.2 | Semi standard non polar | 33892256 | | 11,14,15-THETA,1TMS,isomer #2 | CCCCCC(O)C(/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C | 3005.6 | Semi standard non polar | 33892256 | | 11,14,15-THETA,1TMS,isomer #3 | CCCCCC(O)C(O)/C=C/C(C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C | 3042.1 | Semi standard non polar | 33892256 | | 11,14,15-THETA,1TMS,isomer #4 | CCCCCC(O)C(O)/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C | 2921.2 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TMS,isomer #1 | CCCCCC(O[Si](C)(C)C)C(/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C | 2973.0 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TMS,isomer #2 | CCCCCC(O[Si](C)(C)C)C(O)/C=C/C(C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C | 2996.1 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TMS,isomer #3 | CCCCCC(O[Si](C)(C)C)C(O)/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C | 2897.9 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TMS,isomer #4 | CCCCCC(O)C(/C=C/C(C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C)O[Si](C)(C)C | 3002.5 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TMS,isomer #5 | CCCCCC(O)C(/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 2922.1 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TMS,isomer #6 | CCCCCC(O)C(O)/C=C/C(C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 2943.2 | Semi standard non polar | 33892256 | | 11,14,15-THETA,3TMS,isomer #1 | CCCCCC(O[Si](C)(C)C)C(/C=C/C(C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C)O[Si](C)(C)C | 2944.0 | Semi standard non polar | 33892256 | | 11,14,15-THETA,3TMS,isomer #2 | CCCCCC(O[Si](C)(C)C)C(/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 2880.7 | Semi standard non polar | 33892256 | | 11,14,15-THETA,3TMS,isomer #3 | CCCCCC(O[Si](C)(C)C)C(O)/C=C/C(C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 2876.8 | Semi standard non polar | 33892256 | | 11,14,15-THETA,3TMS,isomer #4 | CCCCCC(O)C(/C=C/C(C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C)O[Si](C)(C)C)O[Si](C)(C)C | 2892.3 | Semi standard non polar | 33892256 | | 11,14,15-THETA,4TMS,isomer #1 | CCCCCC(O[Si](C)(C)C)C(/C=C/C(C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C)O[Si](C)(C)C)O[Si](C)(C)C | 2828.2 | Semi standard non polar | 33892256 | | 11,14,15-THETA,1TBDMS,isomer #1 | CCCCCC(O[Si](C)(C)C(C)(C)C)C(O)/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O | 3240.2 | Semi standard non polar | 33892256 | | 11,14,15-THETA,1TBDMS,isomer #2 | CCCCCC(O)C(/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C(C)(C)C | 3250.5 | Semi standard non polar | 33892256 | | 11,14,15-THETA,1TBDMS,isomer #3 | CCCCCC(O)C(O)/C=C/C(C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C(C)(C)C | 3277.1 | Semi standard non polar | 33892256 | | 11,14,15-THETA,1TBDMS,isomer #4 | CCCCCC(O)C(O)/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C(C)(C)C | 3165.1 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TBDMS,isomer #1 | CCCCCC(O[Si](C)(C)C(C)(C)C)C(/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C(C)(C)C | 3441.5 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TBDMS,isomer #2 | CCCCCC(O[Si](C)(C)C(C)(C)C)C(O)/C=C/C(C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C(C)(C)C | 3451.3 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TBDMS,isomer #3 | CCCCCC(O[Si](C)(C)C(C)(C)C)C(O)/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C(C)(C)C | 3392.1 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TBDMS,isomer #4 | CCCCCC(O)C(/C=C/C(C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 3463.2 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TBDMS,isomer #5 | CCCCCC(O)C(/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 3416.4 | Semi standard non polar | 33892256 | | 11,14,15-THETA,2TBDMS,isomer #6 | CCCCCC(O)C(O)/C=C/C(C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 3429.9 | Semi standard non polar | 33892256 | | 11,14,15-THETA,3TBDMS,isomer #1 | CCCCCC(O[Si](C)(C)C(C)(C)C)C(/C=C/C(C/C=C\C/C=C\CCCC(=O)O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 3589.9 | Semi standard non polar | 33892256 | | 11,14,15-THETA,3TBDMS,isomer #2 | CCCCCC(O[Si](C)(C)C(C)(C)C)C(/C=C/C(O)C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 3571.3 | Semi standard non polar | 33892256 | | 11,14,15-THETA,3TBDMS,isomer #3 | CCCCCC(O[Si](C)(C)C(C)(C)C)C(O)/C=C/C(C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 3606.8 | Semi standard non polar | 33892256 | | 11,14,15-THETA,3TBDMS,isomer #4 | CCCCCC(O)C(/C=C/C(C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 3619.0 | Semi standard non polar | 33892256 | | 11,14,15-THETA,4TBDMS,isomer #1 | CCCCCC(O[Si](C)(C)C(C)(C)C)C(/C=C/C(C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 3740.0 | Semi standard non polar | 33892256 |
|
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - 11,14,15-THETA GC-MS (Non-derivatized) - 70eV, Positive | splash10-0udr-5593000000-b44c164b1afc4290e62a | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 11,14,15-THETA GC-MS (4 TMS) - 70eV, Positive | splash10-00b9-8310987000-e42844df87dbeaaac7b7 | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 11,14,15-THETA GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 10V, Positive-QTOF | splash10-00kr-0019000000-5d98b30fc798f154e3cd | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 20V, Positive-QTOF | splash10-0600-8697000000-f2898f6e9aa37eb19701 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 40V, Positive-QTOF | splash10-05tf-9420000000-4b2f1a462946904efa65 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 10V, Negative-QTOF | splash10-0udi-0009000000-81208c2a7c484c87062f | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 20V, Negative-QTOF | splash10-0f79-4389000000-3a054cf28c217520eeed | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 40V, Negative-QTOF | splash10-0a4i-9340000000-315051dfef85d54b3de6 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 10V, Positive-QTOF | splash10-00kr-0119000000-54253aeda6e4903c01bc | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 20V, Positive-QTOF | splash10-014r-4339000000-fe248a9e503f326b0181 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 40V, Positive-QTOF | splash10-05nf-9400000000-b63770838f1639e0172e | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 10V, Negative-QTOF | splash10-0udi-0019000000-26292874068f05134a76 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 20V, Negative-QTOF | splash10-0kbs-3967000000-5212de307caf1623f831 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 11,14,15-THETA 40V, Negative-QTOF | splash10-0002-9530000000-88a46328b37a9b5888f5 | 2021-09-23 | Wishart Lab | View Spectrum |
|
|---|